Conjugated acrilonitriles: minimalist structures with leishmanicida activity
Leishmaniasis is one of the most devastating tropical zoonotic diseases, with an estimated incidence of around one million new cases per year. It is transmitted to humans through the bite of the female of a family of hematophagous insects (mosquitoes), the so-called sandflies, which are infected by the protozoan Leishmania and act as its biological vector. The most common clinical manifestation of this disease is cutaneous Leishmaniasis, which manifests itself in the form of ulcerative lesions in exposed areas of the body, which can leave scars for life. From the pharmacological point of view, the treatments in use continue to be the same as in the 20th century, with long therapeutic times, limited effectiveness and high toxicity. Although in recent years there has been an increase in activity in the search for new therapies, the development of a truly effective pharmacological response requires the systematic exploration of its pharmacological space and the discovery of new chemotypes with potential therapeutic activity.
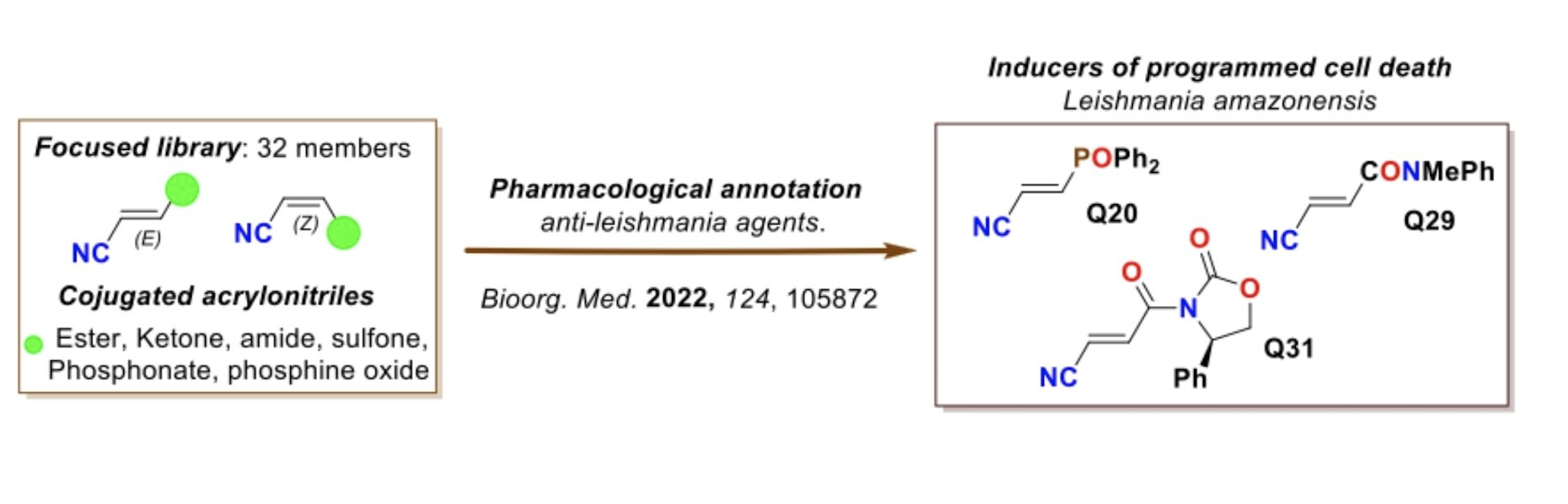
In this line of exploration and search, the research teams of the Instituto de Productos Naturales and Agrobiología (IPNA-CSIC) (Drs. Tejedor Aragón and García Tellado; Molecular Sciences Department; Molecular Structure, Design and Function research group) and the Instituto de Enfermedades Tropicales y Salud Pública de las Islas Canarias (ULL), led by doctors Lorenzo-Morales and Piñero Barroso, are developing a collaborative research project aimed at discovering new synthetic chemotypes with potential therapeutic use against this disease. The result of this effort has been the discovery of a new chemotype based on the acrylonitrile unit (see figure), a minimalist molecular structure with proven activity against the disease known as Chagas disease1. In this study, the authors describe a new pharmacological annotation for these chemotypes and report their therapeutic potential as leishmanicidal agents.
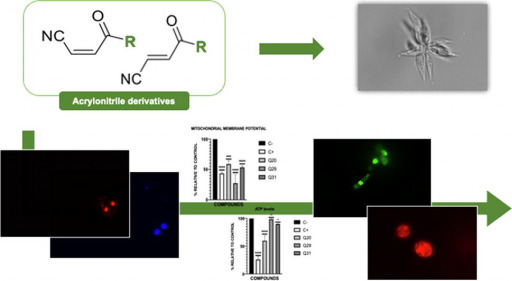
For this study, a library of 32 conjugated acrylonitriles was tested in vitro against Leishmania amazonensis (see figure). Three members of the library, Q20 (12.41), Q29 (11.2) and Q31 (11.56), showed better selectivity than miltefosine (11.14; the reference standard agent) against promastigotes (the form of the parasite in mosquitoes and culture media) of these parasites. The study of their mechanisms of action showed that the three acrolinitriles produce chromatin condensation, decreased mitochondrial membrane potential, altered plasma permeability, and production of reactive oxygen species. Overall, these events seem to indicate that these acrylonitriles induce programmed cell death. The results of this study have been published as an open access article in Bioorganic Chemistry.
C. J. Bethencourt-Estrella, S. Delgado-Hernández, A. López-Arencibia, D. San Nicolás-Hernández, D. Tejedor, F. García-Tellado, J. Lorenzo-Morales, J. E. Piñero. In vitro activity and cell death mechanism induced by acrylonitrile derivatives against Leishmania amazonensis. Bioorganic Chemistry 2022, 124, 105872. DOI: 10.1016/j.bioorg.2022.105872.
1C. J. Bethencourt-Estrella, S. Delgado-Hernández, A. López-Arencibia, D. San Nicolás-Hernández, I. Sifaoui, D. Tejedor, F. García-Tellado, J.Lorenzo-Morales, J. E. Piñero. Acrylonitrile Derivatives against Trypanosoma cruzi: In Vitro Activity and Programmed Cell Death Study. Pharmaceuticals 2021, 14, 552.